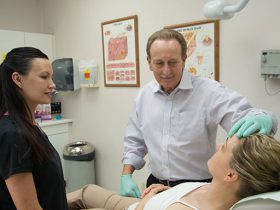
There are easy-to-follow ways to make one’s dermatological visit go smoother and quicker Visiting a state-of-the-art dermatological facility has become quite commonplace. In 2018, an impressive 16.04% of people aged 18 to 29 visited a dermatologist, while 12.91% of those between the ages of 30 to 49 did so. A full 15.73% of those aged … Read more › “Dr. F. Victor Rueckl of Lakes Dermatology Offers Some Helpful Pre-Appointment Tips”
JUVÉDERM® VOLUMA™ XC IS FIRST AND ONLY PRODUCT OF ITS KIND APPROVED BY THE FDA FOR USE IN THE CHIN
IRVINE, Calif., June 15, 2020 /PRNewswire/ — Today Allergan Aesthetics, an AbbVie company (NYSE: ABBV), announced the FDA approval of JUVÉDERM® VOLUMA™ XC for the augmentation of the chin region to improve the chin profile in adults over the age of 21.1 As the category leader, the JUVÉDERM® Collection of Fillers has the broadest portfolio of specifically tailored treatment options, and this … Read more › “JUVÉDERM® VOLUMA™ XC IS FIRST AND ONLY PRODUCT OF ITS KIND APPROVED BY THE FDA FOR USE IN THE CHIN”
New FDA-Approved Kybella Melts Away the Double Chin Notes Dr. F. Victor Rueckl of Lakes Dermatology
Men are flocking to dermatologists to take advantage of Kybella Most individuals who seek to maintain a youthful appearance tend to pay particular attention to the face, such as the areas around the eyes and mouth. A lot of concern is focused on combating the fine lines and wrinkles that will pop up as one … Read more › “New FDA-Approved Kybella Melts Away the Double Chin Notes Dr. F. Victor Rueckl of Lakes Dermatology”
Dr. F. Victor Rueckl of Lakes Dermatology Commends Consumer Reports; Study on Physical and Chemical Sunscreens
Chemical sunscreens are proven to provide better protection The summer season is almost here, much to the joy of schoolchildren everywhere. Summer brings a lot of fun activities: fishing, backyard grilling, picnics, family vacations, trips to the beach, and so on. Another feature of summer that most older people are familiar with is sunburn. In … Read more › “Dr. F. Victor Rueckl of Lakes Dermatology Commends Consumer Reports; Study on Physical and Chemical Sunscreens”
GETTING CORRECT DIAGNOSIS IS FIRST STEP IN DEALING WITH DISCOMFORT OF ECZEMA
By LINDA J. SIMPSON SPECIAL TO THE LAS VEGAS REVIEW-JOURNAL Eczema has been called “the itch that rashes.” The terms eczema and dermatitis are often used interchangeably to describe a reaction pattern of the skin that creates itching, redness and rashes. There are different types of eczema. Many people are familiar with the contact dermatitis … Read more › “GETTING CORRECT DIAGNOSIS IS FIRST STEP IN DEALING WITH DISCOMFORT OF ECZEMA”
An Unforeseen Consequence of the Affordable Care Act Is Losing Access To Dermatologists, According To Dr. F. Victor Rueckl
Switching To Medicare Advantage May Cost Patients’ Access To Their Dermatologist The Affordable Care Act was designed to provide access to medical treatment for every individual. To that end, a grand total of 11.4 million Americans signed up for private health insurance by the end of the second enrollment period. (1) This is tremendous … Read more › “An Unforeseen Consequence of the Affordable Care Act Is Losing Access To Dermatologists, According To Dr. F. Victor Rueckl”